Automating bacterial enumeration
Posted: 21 November 2005 | Roy Betts, Head of Microbiology, Campden & Chorleywood Food Research Association | No comments yet
Quality indicator (QI) tests represent the large majority of routine tests currently performed by food microbiology laboratories. Although not necessarily pathogenic, indicator organisms, such as Total Plate Count, coliforms, Escherichia coli, Enterobacteriaceae, yeasts and moulds can alter the appearance and taste of a product when present in large quantities. The enumeration of the microbial flora present indicates the microbiological quality of the raw materials and the finished product, as well as ensuring in-process control. QI tests are therefore of major importance in establishing a consistent level of hygiene during food production and a good quality product.
Quality indicator (QI) tests represent the large majority of routine tests currently performed by food microbiology laboratories. Although not necessarily pathogenic, indicator organisms, such as Total Plate Count, coliforms, Escherichia coli, Enterobacteriaceae, yeasts and moulds can alter the appearance and taste of a product when present in large quantities. The enumeration of the microbial flora present indicates the microbiological quality of the raw materials and the finished product, as well as ensuring in-process control. QI tests are therefore of major importance in establishing a consistent level of hygiene during food production and a good quality product.
Quality indicator (QI) tests represent the large majority of routine tests currently performed by food microbiology laboratories. Although not necessarily pathogenic, indicator organisms, such as Total Plate Count, coliforms, Escherichia coli, Enterobacteriaceae, yeasts and moulds can alter the appearance and taste of a product when present in large quantities. The enumeration of the microbial flora present indicates the microbiological quality of the raw materials and the finished product, as well as ensuring in-process control. QI tests are therefore of major importance in establishing a consistent level of hygiene during food production and a good quality product.
The ‘labour’ issue related to conventional microbiological testing is well-known and over the past twenty-five years, the field of research into alternative test methods and automated instruments that can decrease test time and improve sample throughput, has grown significantly. However, in contrast to the field of pathogen detection technology, which can now be highly automated, few developments have occurred in QI analysis where conventional methods tend to prevail. QI counts are still mostly determined by labour-intensive tube and plate counting methods, despite the fact that, as a general rule, laboratories test far higher sample numbers of indicator organisms than pathogens.
Advantages of automated QI testing
The presence of large numbers of QI organisms in a food product can significantly impact manufacturers. Specifications may be exceeded, shelf life may be compromised and products may show signs of spoilage when handled by consumers. This can lead to a fall in the market value of the product, which will fail to sell well in supermarket displays, or may rapidly become unfit for consumption. More seriously, all of these outcomes may lead to product rejection, product return and loss of credibility for the manufacturer. QI tests help manufacturers ensure the microbiological quality of the product and if carried out quickly and efficiently, reduce the likelihood of rejection and return from consumers and retail stores.
Automated methods for QI testing are therefore of interest to the food industry, which is constantly driven to increase efficiency and productivity and, in particular, to high-volume food microbiology laboratories.
Automation can provide significant advantages to food laboratories, including higher throughput and full traceability that can all result from the implementation of well-researched and validated automated methods. This can lead to laboratory staff having greater time for value added tasks, becoming more proactive and anticipating problems and issues both in the laboratory and factory, rather than reacting to problems that have occurred.
The technical challenges
The development of an automated QI enumeration system involves several technical challenges:
- Responding to food laboratory requirements to perform a high volume of tests per person per day – the demand for QI tests in the food industry is constantly increasing
- Offer an equivalent or superior level of performance to that obtained with manual test methods, but with added benefits such as the standardisation of the preparation and reading processes and the transfer of data
Assessment of an automated solution for QI enumeration
A very limited number of automated QI test systems have been developed in the past. These have been based on a number of technologies such as impedance/ conductance microbiology, automated colorimetry and flow cytometry. Very recently, a new approach to the automation of QI tests has been developed.
Campden & Chorleywood Food Research Association (CCFRA) has recently performed a 2-month evaluation of this new method, which aims to automate and simplify existing manual methods. The objective of the evaluation was to compare the performance of the automated system with the ISO reference plate count methods.
Automated system principle
TEMPO® is an automated system based on traditional QI enumeration methods. The system design replaces serial dilutions, media preparation and plate reading with a 1/10 dilution and automated enumeration based on the traditional and accepted MPN (Most Probable Number) method, miniaturised on a card format.
The TEMPO system (bioMérieux) consists of two parts: a Preparation Station and a Reading Station. The system comprises two single-use disposables: a vial containing dehydrated culture medium and an enumeration card that miniaturises the MPN method, using three series of 16 wells each. The culture medium, specific for the microbial group to be enumerated, is adapted to ensure rapid detection of microbial growth through detection of changes in fluorescence. After inoculation with a dilution of the food sample, the culture medium is automatically transferred into the card by the TEMPO Filler. The card is then incubated in a normal incubator. During incubation, microbial growth causes a modification of the substrate in the medium, revealed through a fluorescent signal that is automatically detected by the TEMPO Reader. Depending on the number of positive wells on the card, the system calculates the number of microorganisms present in the sample with a range of 1-9.8 x 107 CFU/g according to the dilution of sample used.
Evaluation protocol
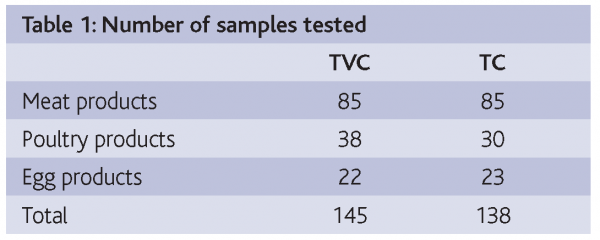
Fresh, frozen and dehydrated meat, poultry and egg products were used to evaluate the following TEMPO tests: TVC (Total Viable Count) and TC (Total Coliforms).
Samples were collected from retail outlets. Ten grams were homogenised in 90 ml of MRD (primary 1/10 dilution) in the specific TEMPO bag with filter and analysed by TEMPO and ISO standard methods in parallel.
Automated method protocol
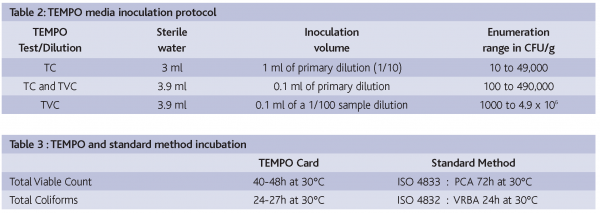
The TEMPO media were re-hydrated with sterile water and inoculated with an aliquot of the homogenised sample (Table 2). The TEMPO system was then used to perform the automated filling of the test card. The instrument then monitored the incubation time of the test cards (Table 3) and, following the appropriate incubation time, alerted the laboratory staff that the cards needed to be read. The cards were analysed in the reader instrument and the final result was then automatically calculated.
Standard method protocol
1 ml of primary dilution and serial dilutions up to 10-3 in MRD were prepared; 3 dilutions for Total Viable Count and 2 dilutions for Total Coliforms; 2 plates per dilution. After inoculation and incubation (Table 3), colonies were counted by visual inspection.
Results for TVC
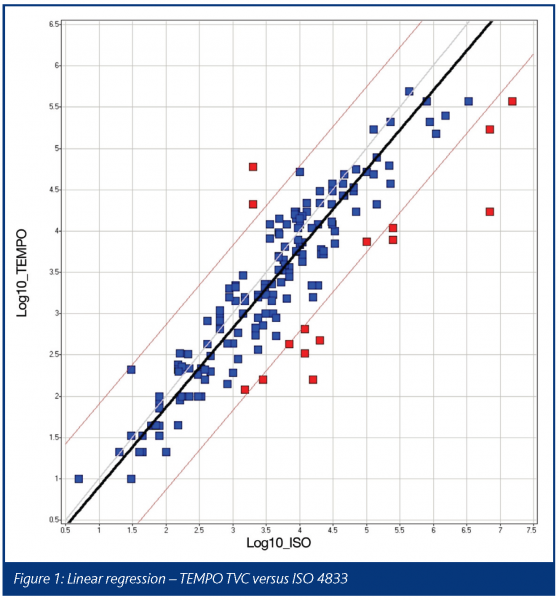
The results obtained for the TVC tests were analysed by linear regression and this is shown in Figure 1.
Generally, there is a good correlation between the results obtained with the standard method and those obtained with TEMPO, with a correlation coefficient of 0.91. The correlation line would indicate that there is almost equivalence between the results, with TEMPO giving a slight positive bias of approximately 0.15 to 0.25 log over the standard method across the range of counts obtained in this study.
Results for coliforms
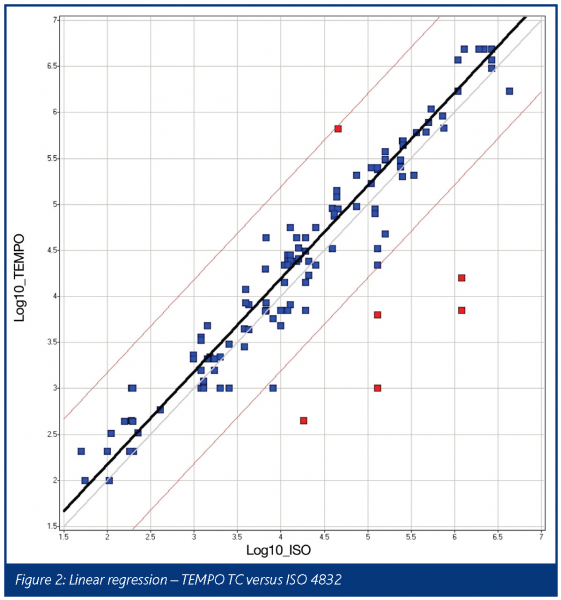
The results obtained for the coliform tests were also analysed by linear regression and this is shown in Figure 2.
Again, a good correlation was obtained between the standard method and the TEMPO method with a correlation coefficient of 0.93. The correlation line indicates a close equivalence between the two methods with TEMPO giving a very slight negative bias of between 0.1 and 0.25 log over the standard method across the range of counts obtained in this study.
Conclusions
Overall, considering that the standard method is based on a manual plate count and the TEMPO method on an automated MPN approach, the results obtained for both TVC and coliform tests compare very well. Not only is there a good correlation for both analyses, but there appears to be very little bias in results.
The food industry places much significance on the results of Quality Indicator tests and is performing a very large number of such tests on a daily basis. The availability of automated methods capable of performing such tests will help parts of the industry by providing a standardised approach and enabling an increase in sample throughput.
This work has shown that TEMPO can offer an automated approach to Quality Indicator testing and provide results comparable to standard plating methods for TVC and coliform testing.