From laboratory-scale to pilot-scale
Posted: 4 November 2010 | Ariette Matser & Hennie Mastwijk, Wageningen UR and Milan Houška, Food Research Institute Prague | No comments yet
The implementation of a novel processing technology needs a science-based approach where product benefits initially demonstrated in a laboratory environment and the associated risks are used to predict enhanced quality when the technology is used in large-scale industrial operation. We discuss four novel technologies business cases developed for food application moving from laboratory to industrial-scale application.
The implementation of a novel processing technology needs a science-based approach where product benefits initially demonstrated in a laboratory environment and the associated risks are used to predict enhanced quality when the technology is used in large-scale industrial operation. We discuss four novel technologies business cases developed for food application moving from laboratory to industrial-scale application.
The implementation of a novel processing technology needs a science-based approach where product benefits initially demonstrated in a laboratory environment and the associated risks are used to predict enhanced quality when the technology is used in large-scale industrial operation. We discuss four novel technologies business cases developed for food application moving from laboratory to industrial-scale application.
For the successful implementation of novel processing schemes – working back from product launch – it is important for reliable equipment to be available at laboratory and pilot-scales. Validation of products and processes on an industrial scale is less thorough than pilot-scale testing: smaller batch sizes allow more critical parameters to be assessed, resulting in a better understanding of the process. In many cases, large-scale industrial equipment cannot be used; for example, laboratory-scale equipment is indispensible for checking critical parameters related to microbial inactivation kinetics of targeted pathogens.
The availability of product specific data for novel processing is a significant problem for risk assessment, which nevertheless has to be achieved prior to the introduction of new products in the European food and beverage markets. Within the 6th Framework of the European Commission, the Integrated Project NovelQ has examined specific research and development issues that create problems in scaling up laboratory equipment to pilot and full-scale industrial units. Topics covered include both generic issues such as the effects on homogeneity when volume-to-surface ratios are changed, as well as practical solutions for de-mixing phenomena. Usually, the development of large, industrial-scale novel technologies is triggered by feasibility studies using small, laboratory-scale equipment where small sample numbers and volumes can be evaluated. When supporting evidence indicates the process can enhance quality and / or shelf life of products, processes are scaled-up to evaluate small numbers of realistic food products (pilot-scale) and industrial-scale equipment developed where these studies are positive. At each stage, specific problems encountered have to be evaluated and resolved before moving to larger-scale production.
From our analysis, we surmise that prior to large-scale utilisation of a technology, it is important that:
» Laboratory-scale equipment should be available for trials with model systems and to build a sound scientific base describing physical-chemical reactions, inactivation kinetics of pathogens, spoilage microorganisms and enzymes
» Small-scale equipment should be located in microbiological and chemical laboratories for experiments with specific components (e.g. pathogens)
»Pilot-scale equipment should be accessible to perform small trials with real food matrices (products) in likely final packaging to study the effect of processing conditions on shelf life and quality, and establish any implications, seen and unforeseen. Typically, pilot-scale systems are used for feasibility and demonstration activities
» Industrial-scale testing equipment, including pre- and post-processing facilities as required, is essential for proper implementation of the process in the industry
It is fundamental that equipment is fully characterised to ensure control over the results and allow proper comparison with products from other systems. Only under such conditions can the small and pilot-scale results translate into specifications for industrial units. Within NovelQ, research and development and scaling-up activities were focused on equipment for high pressure pasteurisation and sterilisation, pulsed electric field processing, cold plasma treatment and advanced heating.
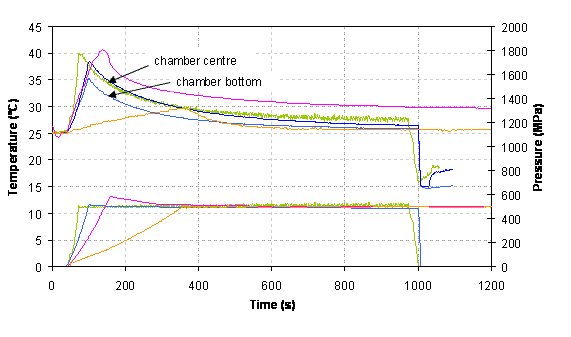

Figure 1 Example of pressure (lower part of graph) and temperature (upper part) profiles during HP treatment at various research units
Up-scaling principles
The principles for scaling-up have to be determined for each technology separately. For high-pressure pasteurisation treatment, equipment is available at all relevant scales within the NovelQ consortium. The activities of equipment suppliers, universities and research organisations have led to reliable industrial units for food manufacturers. Because high-pressure pasteurisation is widely used, experience is available concerning maintenance and operational efficiency, and insights have been shared with (potential) users.
For thermally-assisted high-pressure sterilisation treatment, reliability of equipment at larger scales is a serious problem. Due to the high temperatures used in combination with high pressures – allowing inactivation of spores (sterilisation regime) – current designs of high-pressure pasteurisation units cannot be used. Laboratory and pilot-scale units are available, but not industrial units. More in-depth research and development is needed for vessel design, sealing and homogeneity at high pressure and high temperature. Research in NovelQ has shown that design and selection of materials is crucial for achieving proper sealing at, for example, 700 MPa and 115°C. Although high-pressure sterilisation is a combination of high-pressure and high-temperature processes, homogeneity of temperature distribution in the vessel during the process is just as important as it is with other thermal processes.
At the start of NovelQ, pulsed electric field equipment was available for pasteurisation of pumpable fluids on a scale of several hundred litres per hour. This flow-through capacity is sufficient for performing pilot studies with real food products. The most important scaling issue, however, relates to chamber design to ensure the treatment of fluids in a homogeneous manner without generating excess heat; fluids may contain small-sized particles up to one centimetre in diameter. Integration of PEF into production lines is also of concern with respect to pre and postprocessing aseptic packaging, as is hygiene and contamination of the entire production line, which is essential to guarantee safe production at reduced temperatures. These issues are not normally considered with pilot-scale equipment. Periodic in-line cleaning and presterilisation of PEF units were found to have a significant impact on the process and chamber design. However, expertise gained in NovelQ means many of these hurdles have now been overcome.
Scaling-up of advanced heating tech – nologies was only investigated in part by NovelQ. Special focus was placed on the interaction between the impact of volume on quality and new menu designs. In general, industrialscale equipment is available for technologies such as ohmic heating, microwave and radio frequency heating, defrosting and drying.
Characterisation of equipment
When evaluating the potential for novel processing in the food industry, companies often perform feasibility studies at laboratoryand pilot-scales. It is important for these experiments to be performed in a consistent and harmonised manner to ensure the results can be compared and judged objectively. It is crucial that results from scientific literature and professional journals are considered, allowing the pros and cons to be evaluated fully, and science-based information presented to consumers and legislators. The global harmonisation initiative (www.globalharmonization.org; supported by NovelQ’s partners EFFoST) is striving for validated and reliable sources, based on scientific consensus and reproducible experiments.
NovelQ developed standard operating procedures to ensure partners used the same research protocols for test equipment. These have been thoroughly tested in practice for high-pressure equipment – operated over a wide range of processing conditions – and are available in the laboratories of many NovelQ partners.
Figure 1 shows the temperature of pressure transmitting media and pressure profiles of four different high pressure units during a standard experiment at 500 MPa and 25°C starting temperature. The graphs reveal the wide range of conditions arising with real products. The main differences observed include the pressure-building time and the impact of vessel dimensions, especially the ratio between vessel height and diameter, pressure fluids and design of the units (e.g. use of internal or external intensifier for pressure). Thus, characterising the equipment used and comparing equipment conditions at laboratory and pilot-scales with industrial-scale units is imperative.
Development of specific research units
Easy access to hands-on support and the opinions of independent experts substantially increases the success of novel technologies in industry. Sharing experiences in a research network such as the NovelQ Industry Advisory Platform (IAP) yields convincing arguments for investment strategies uninhibited by issues such as confidentiality. Small-scale units for high-pressure, pulsed electric field and advanced heating technologies are already available. Cold plasma, however, is a more recent innovation for non-chemical surface decontamination. Within NovelQ, this potential was recognised at an early stage and, consequently, the need for standardised laboratory-scale units generating food-grade cold plasmas. With such a unit, research organisations and universities can perform relevant studies on, for example, inactivation kinetics of micro-organisms. A wellcharacterised small-scale unit was developed for the production of nitrogen-based plasma gas (see Figure 2). Initial decontamination tests successfully carried out by Wageningen UR Food & Biobased Research (NL) and the Institute of Food Research (UK) produced identical results. These units are currently being used for pathogen studies, related to food safety issues, as well as studies on food quality. Such developments will improve knowledge of cold plasma applications and support implementation of this technology by the food industry.


Figure 2 Characteristic lab-scale HP equipment for inactivation kinetics (multi-vessel system, Wageningen UR)
Availability and accessibility of pilot-scale equipment for HPP, PEF and advanced heating with detailed characteristics
Pilot-scale equipment, available at some NovelQ partners, has enhanced implementation of novel technologies, (see Table 1). One and two-day trials were organised during the project, allowing food manufacturers to test their products using pilot facilities whilst evaluation of the processing conditions on quality and shelf life were carried out jointly. If New Food readers are interested in evaluating the effects of a specific novel process on their products, they are welcome to contact the relevant NovelQ partner.
Table 1: Pilot Equipment at NovelQ Partners
| NovelQ partner | Characteristics | Location | |||
| High pressure processing | Volume [l] | Pressure [MPa] | T [°C] | ||
| Wageningen UR, Food & Biobased Research | 4 | 800 | 125 | Wageningen, The Netherlands | |
| TU Berlin, Department of Food Biotechnology and Food Process Engineering | 0.75 | 800 | 80 | Berlin, Germany | |
| KU Leuven, Laboratory of Food Technology | 0.6 | 690 | 100 | Leuven, Belgium | |
| SIK | 1.4 | 900 | 80 | Gothenburg, Sweden | |
| TNO Quality of Life | 2 | 900 | 60 | Zeist, The Netherlands | |
| Food Research Institute Prague | 2 | 550 | 60 | Prague, Czech Republic | |
| Beskyd Frycovice a.s. via Food Research Institute Prague | 125 | 450 | ambient | Frycovice, Czech Republic | |
| Friedrich Alexander Universität Erlangen Nürnberg | 2 | 700 | 80 | Erlangen, Germany | |
| NC Hyperbaric | 55 | 600 | 30 | Burgos, Spain | |
| Pulsed electric field processing | Capacitiy [l/h] | ||||
| Wageningen UR, Food & Biobased Research | 200 | Wageningen, The Netherlands | |||
| TU Berlin, Department of Food Biotechnology and Food engineering | 600 | Berlin, Germany | |||
| University of Lleida | 28 | Lleida, Spain | |||
| University of Zaragoza | 20 | Zaragoza, Spain | |||
| Keki | 7.2 | Budapest, Hungary | |||
| Advanced heating | Technology | Power [kW] | |||
| Wageningen UR, Food & Biobased Research | Microwave | 6 | Wageningen, The Netherlands | ||
| Wageningen UR, Food & Biobased Research | Radio frequency | 7 | Wageningen, The Netherlands | ||
| TU Berlin, Department of Food Biotechnology and Food Process Engineering | Ohmic | 11.5 | Berlin, Germany | ||
| CTCPA | Ohmic | Avignon, France | |||
| Opal | Ohmic | 10-50 | France | ||
About the Authors
Ariette Matser
Ariette Matser is currently Senior Scientist – Mild Preservation and Novel Technologies and Programme Coordinator – Mild Preservation within Food & Biobased Research of Wageningen UR. She studied Food Science at Wageningen UR. Her primary research activities include novel technologies and food safety and quality, preservation and processing of food products and effects on food quality, and food structure and the effects of processing on this. Management activities are involvement in (inter) national R&D projects, project acquisition and coordination of activities on novel technologies. She is Project Coordinator of the European project NovelQ.
Hennie Mastwijk
Hennie Mastwijk is a senior scientist at Wageningen UR Food & Biobased Research, The Netherlands (www.wur.nl). He is involved in the development of equipment and food products through electronic based mild preservation processes.
Milan Houška
Milan Houška is the head of the department of food engineering at the Food Research Institute Prague. He is dealing with physical properties of foods and heat and novel processes including their mathematical modelling. He is the author of many scientific papers, patents and innovations already marketed. He coordinated the project that finalised with high pressure pasteurised juices on the Czech food market.




